ISO 13485 Medical Devices Quality Management System – Lead Auditor Training Courses
What is ISO 13485?
As an international standard, ISO 13485 determines the requirements for the medical devices industry. This standard was established to be used by companies during the life cycle of medical devices, from the production to post-production, including decommission and disposal.
However, ISO 13485 can also be operated by other parties such as certification bodies which can assist in the certification processes. ISO 13485 serves as a tool for organizations to develop and retain their processes efficiently. Organizations that are striving for continuous improvement will benefit from this standard by being able to provide safe and competent medical devices and achieve the trust of customers.
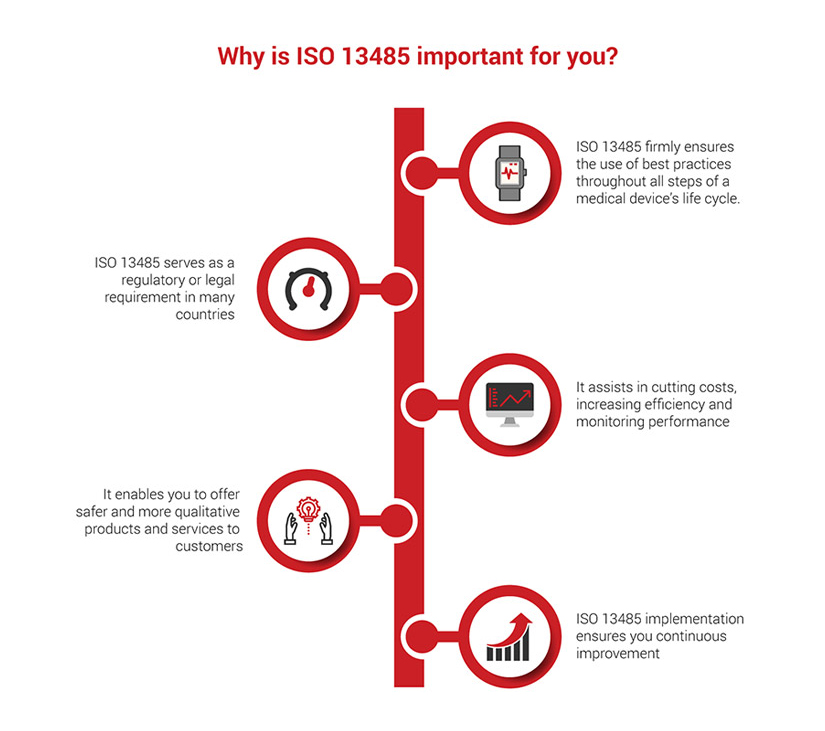
Why is Medical Devices Quality Management System important for you?
Being an ISO 13485 certified professional imposes your determination to offer qualitative products and services to your company and/or clients. Attaining the necessary knowledge and skills to operate an ISO 13485 framework demonstrates your commitment to helping your company ensure continuous improvement and better work processes.
It also indicates that you understand the importance of the safety and performance of medical devices and how a medical devices quality management system can assure just that, consequently leading to customer satisfaction. Similarly, it can introduce you to new opportunities, such as working for large companies that offer quality products and services. These companies will value your knowledge and expertise on this standard, while enabling you to maximize your earning potential.
Benefits of ISO 13485 Medical Devices Quality Management System
By becoming an ISO 13485 certified professional, you will be able to:
- Increase the number of job opportunities
- Create new business connections
- Gain competitive advantage
- Increase effectiveness and lower costs
- Offer expertise on improving work processes
- Offer safer and more efficient medical devices
- Increase customer satisfaction
ISO 13485 Lead Auditor
ISO 13485 Lead Auditor
ISO 13485 Lead Auditor training enables you to develop the necessary expertise to perform a Medical Devices Quality Management System (MDQMS) audit by applying widely recognized audit principles, procedures, and techniques.
Why should you attend?
During this training course, you will acquire the knowledge and skills to plan and carry out internal and external audits in compliance with ISO 19011 and ISO/IEC 17021-1 certification process.
Based on practical exercises, you will be able to master audit techniques and become competent to manage an audit program, audit team, communication with customers, and conflict resolution.
After acquiring the necessary expertise to perform this audit, you can sit for the exam and apply for a “PECB Certified ISO 13485 Lead Auditor” credential. By holding a PECB Lead Auditor Certificate, you will demonstrate that you have the capabilities and competencies to audit organizations based on best practices.
Who should attend?
- Auditors seeking to perform and lead Medical Devices Quality Management System (MDQMS) certification audits
- Managers or consultants seeking to master a Medical Devices Quality Management System audit process
- Individuals responsible for maintaining conformance with Medical Devices Quality Management System requirements
- Technical experts seeking to prepare for a Medical Devices Quality Management System audit
- Expert advisors in Medical Devices Quality Management
Learning Objectives
- Understand the operations of a Medical Devices Quality Management System based on ISO 13485
- Acknowledge the correlation between ISO 13485 and other standards and regulatory frameworks
- Understand an auditor’s role to: plan, lead and follow-up on a management system audit in accordance with ISO 19011
- Learn how to lead an audit and audit team
- Learn how to interpret the requirements of ISO 13485 in the context of a MDQMS audit
- Acquire the competencies of an auditor to: plan an audit, lead an audit, draft reports, and follow-up on an audit in compliance with ISO 19011
Educational approach
- This training is based on both theory and best practices used in MDQMS audits
- Lecture sessions are illustrated with examples based on case studies
- Practical exercises are based on a case study which includes role playing and discussions
- Practice tests are similar to the Certification Exam
Prerequisites
A fundamental understanding of ISO 13485 and comprehensive knowledge of audit principles.
Course Agenda
-
Day 1: Introduction to Medical Devices Quality Management Systems (MDQMS) and ISO 13485
Day 2: Audit principles, preparation and launching of an audit
Day 3: On-site audit activities
Day 4: Closing the audit
Day 5: Certification Exam
Examination
-
This course is taught in partnership with PECB. The “PECB Certified ISO 13485 Lead Auditor” exam fully meets the requirements of the PECB Examination and Certification Programme (ECP). The exam covers the following competency domains:
Domain 1: Fundamental principles and concepts of a Medical Devices Quality Management System (MDQMS)
Domain 2: Medical Devices Quality Management System (MDQMS)
Domain 3: Fundamental audit concepts and principles
Domain 4: Preparation of an ISO 13485 audit
Domain 5: Conducting an ISO 13485 audit
Domain 6: Closing an ISO 13485 audit
Domain 7: Managing an ISO 13485 audit program
For specific information about exam type, languages available, and other details, please visit the List of PECB Exams and the Examination Rules and Policies.
Certification
-
After successfully completing the exam, you can apply for the credentials shown on the table below. You will receive a certificate once you comply with all the requirements related to the selected credential. For more information about ISO 13485 certifications and the PECB certification process, please refer to the Certification Rules and Policies.
The requirements for PECB Auditor Certifications are:
Credential Exam Professional experience MS audit/assessment experience Other requirements PECB Certified ISO 13485 Provisional Auditor PECB Certified ISO 13483 Lead Auditor Exam or equivalent None None Signing the PECB Code of Ethics PECB Certified ISO 13485 Auditor PECB Certified ISO 13483 Lead Auditor Exam or equivalent Two years: One year of work experience in Medical Devices Quality Management Audit activities: a total of 200 hours Signing the PECB Code of Ethics PECB Certified ISO 13485 Lead Auditor PECB Certified ISO 13483 Lead Auditor Exam or equivalent Five years: Two years of work experience in Medical Devices Quality Management Audit activities: a total of 300 hours Signing the PECB Code of Ethics PECB Certified ISO 13485 Senior Lead Auditor PECB Certified ISO 13483 Lead Auditor Exam or equivalent Ten years: Seven years of work experience in Medical Devices Quality Management Audit activities: a total of 1,000 hours Signing the PECB Code of Ethics Note: PECB Certified Individuals who do possess the Lead Implementer and Lead Auditor Credentials are qualified for the respective PECB Master Credential, given they have taken 4 additional Foundation Exams which are related to this scheme. For more detailed information about the Foundation Exams and the overall Master Requirements, please go to the following link: https://pecb.com/en/master-credentials.
To be considered valid, these audits should follow best audit practices and include the following activities:
- Audit planning
- Audit interview
- Managing an audit program
- Drafting audit reports
- Drafting non-conformity reports
- Drafting audit working documents
- Documentation review
- On-site audit
- Follow-up on non-conformities
- Leading an audit team
General Information
- Certification and examination fees are included in the price of the training course
- Training material containing over 450 pages of information and practical examples will be distributed
- An attestation of course completion worth 31 CPD (Continuing Professional Development) credits will be issued to the participants who have attended the training course.
- In case of exam failure, you can retake the exam within 12 months for free.
For additional information, please contact us at info@globalskillsfactory.fi





